Summer 2022
Updates on committees, bioregistry, research, junior investigators, clinical care, and funding
Note: We’ve updated the format of our newsletter for easier reading on your mobile device. Let us know if you have any comments or questions!
It is with great pleasure that we write to inform you of our latest doings! These last 6 months have been full of momentum, and we are looking forward to continuing the trend. Your enthusiasm and support for the Center for Innovations in Cancer & Transplant provides us with extra energy to push ourselves and the mission to next steps—thank you!
Since we last wrote, we competed in the American Society of Transplantation Visionaries Challenge with a short video introducing CICT, launched several new studies, developed and tested the beta-version of the Bioregistry, and cared for a growing number of Cancer & Organ Transplant Clinic (COTC) patients. In the process, we have had the privilege and joy of welcoming new team members who are future scientists and physicians.
Inaugural meetings of advisory committees
To guide our work, we met with the inaugural CICT Scientific Advisory Committee (SAC) and the Community Engagement Committee (CEC) in May. The CEC will advocate for and publicize CICT to the broader patient and donor community, and ensure that patients’ voices are heard in our research and clinical work. We are delighted that Glenda Roberts of the Kidney Research Institute is chairing this committee. The SAC, chaired by Sue Trinidad of the UW Dept. of Bioethics, will offer guidance on the research and funding agenda while extending the reach of CICT through personal contact. We look forward to meeting with them again in November!
Bioregistry launching soon
A key part of CICT is our bioregistry, which will be the only contemporary bioregistry of solid organ transplant candidates and recipients with cancer in the United States, unique in that it will have patient-level data and, eventually, biological samples for multidimensional research. The registry will integrate research and clinical data from cancer centers and transplant programs, and will catalyze multidimensional and multicenter collaborative research. Soon we will invite collaborators to upload patient data for initial projects. We will incrementally expand the cancer types based on patient benefit. Upon request, we will share data with other investigators.
New research projects
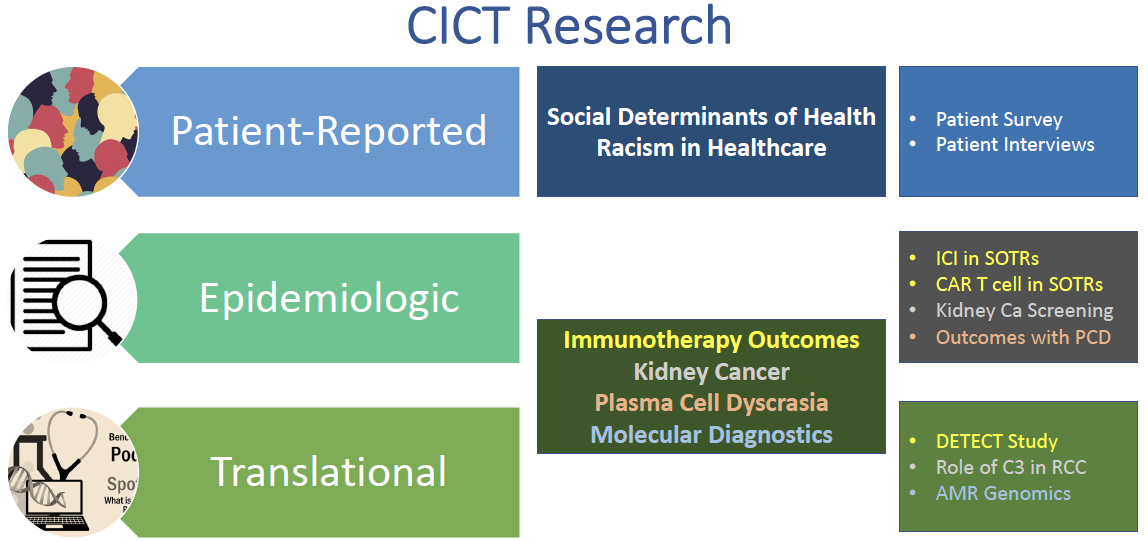
We have also been busy developing and initiating projects within the three dimensions of: Patient-Reported Outcomes, Epidemiologic, and Translational research, with current target areas reflected in the CICT Research figure. However, CICT’s goals relate to all solid organ transplant and all cancers, and as we grow, we will be expanding our research to address other questions.
Role of C3 in Renal Cell Carcinoma
The Role of C3 in renal cell carcinoma (RCC) study formally launched in May. We know that complement C3 is increased in RCC, but its function and significance is unknown. We hope to better understand this in order to identify novel biomarkers and therapies to treat RCC.
DETECT
DETECT asks whether measuring circulating cell-free DNA for kidney transplant rejection and cancer progression provides earlier and more precise evidence of transplant rejection or progressive cancer than standard of care in KTRs treated for advanced cancer. DETECT will begin enrolling this Fall with the goals of minimizing risks and optimizing outcomes.
Equity and justice
Equity and justice in research and clinical care are priorities, and in order to achieve both, we want to understand the roles of racism and discrimination in the patient experience and health outcomes. Our patient survey, led by Drs. Bessie Young and Yue-Harn Ng (UW), is evaluating how social determinants of health and patient perceived racism are linked to health disparities and outcomes in solid organ transplant candidates and recipients, with and without cancer. This study will commence in the fall.
Junior investigators working on additional studies
Involving the next generation of scientists and physicians in CICT is also important to us, and we are delighted to be working with three promising junior investigators on several of our studies!
Alyssa Ong
SUNY Stonybrook graduate
Alyssa Ong has plans for medical school, and worked with Chris during high school. She is helping lead the Kidney Cancer Screening study - a survey of nephrology providers in the US, Australia and New Zealand that assessed current kidney cancer screening practices and views of possible kidney cancer screening guidelines. Despite higher risks, there are no kidney cancer screening guidelines in patients with chronic kidney disease and end-stage kidney disease, including those on dialysis and with a kidney transplant.
Regina Tsay
First-year UW medical student
Regina Tsay is helping to lead a retrospective analysis of outcomes in patients with Plasma Cell Dyscrasia (PCD) who are referred to UW for organ transplant.
Historically, about 50% of patients diagnosed with PCD develop kidney disease but are rarely referred for transplant due to concern for recurrence or progression of cancer after transplant. New therapies for PCD have enabled safer PCD and organ transplant care in the past decade. Yet, we lack contemporary referral and transplant data. We hypothesize a higher prevalence of transplant referrals and better outcomes in patients with PCD in the setting of improved therapies.
Shaaniya Mahabir
Rising UW freshman
Shaaniya Mahabir plans to go to medical school after finishing her undergraduate studies. She is working on our Digital Spacing Profiling (DSP) in Antibody-mediated Rejection (AMR) study. AMR remains the most formidable cause of kidney transplant failure, with limited understanding of the causes and therefore limited diagnostic and therapeutic options. Digital spatial profiling of kidney biopsy specimens is a novel technology that provides spatially defined cell protein/RNA profiles that may help better define causes of AMR and potential therapeutic targets.
Clinical care
Clinical care is a significant complementary element of CICT, and we are busy seeing patients at our COTC, the first of its kind multi-disciplinary consult clinic for organ transplant candidates and recipients with challenging cancer and transplant conditions.
Coordinated, personalized care
We have had the privilege of caring for an increasing number of patients at the COTC since opening at the Fred Hutch Cancer Center (formerly Seattle Cancer Care Alliance) on September 1, 2021. These patients benefit from personalized consultation with relevant cancer and transplant specialists, including unified care recommendations for the patient and referring provider(s). The clinic applies contemporary knowledge and state-of-the-art therapies from 12 cancer specialties and all organ transplant types to address patient-centered issues.
Patient participation in the bioregistry
All patients at COTC will be invited to take part in the CICT bioregistry. The integration of a multidisciplinary clinic and research center provides bidirectional benefits that immediately offers the latest knowledge to the patients while enabling patients to contribute to cutting-edge research in order to improve the future of organ transplant and cancer care.
Funding
Running a center like CICT takes money, and we are happy and hopeful to share some future funding opportunities in the offing!
New opportunities
After a rigorous review, we have been invited to apply for the Department of Defense IDEA Development Award in October to further study the complement system in RCC. In addition, we are collaborating with Canadian colleague Paul Keown and team on an exciting program proposal - Personalized Transplantation, Tissue Engineering and Tolerance.
Deepening your commitment
Grants are just one source of our funding; another significant source is from individual donations, which is where you come in! We invite you to deepen your commitment to CICT and be a part of our work to improve the lives of cancer and transplant patients by making a tax-deductible donation.
We are so grateful, and couldn’t do it without you! Please feel free to contact us with any questions or comments.
Thank you,
Chris Blosser, Barbara Kavanaugh & Caitlin Gard